|
Again, by convention, both Φ and ψ are defined as 180 0 when the polypeptide is in its fully extended conformation and all peptide groups are in the same plane. Why Proteins are Very Important? How to Explain?īy convention, the bond angles resulting from rotations at C α are labeled Φ (phi) for the N-C α bond and ψ (psi) for the C α-C bond.Protein Folding is Made Easy at Cellular Level.The rigid peptide bonds limit the range of conformations that can be assumed by a polypeptide chain. The backbone of a polypeptide chain can thus be pictured as a series of rigid planes, with consecutive planes sharing a common point of rotation at C α. Rotation is permitted about the N-C α and the C α -C bonds. The six atoms of the peptide group lie in a single plane, with the oxygen atom of the carbonyl group and the hydrogen atom of the amide nitrogen Trans to each other.įrom these findings, Pauling and Corey concluded that the peptide C-N bonds are unable to rotate freely because of their Partial double-bond character. The oxygen has a partial negative charge and the nitrogen has a partial positive charge, setting up a small electric dipole. This indicated a resonance or partial sharing of two pairs of electrons between the carbonyl oxygen and the amide nitrogen. Physio Chemical Properties of Amino acids? (Guide).What is Amino acid and its Structural Chemistry?.The peptide C-N bond is somewhat shorter than the C-N bond in a simple amine, and the atoms associated with the peptide bond are coplanar, according to X-ray diffraction investigations of crystals of amino acids, simple dipeptides, and tripeptides. The α-carbons of adjacent amino acid residues are separated by three covalent bonds, arranged as C α-C-N-C α. They began with a careful analysis of the peptide bond. In the late 1930s, Linus Pauling and Robert Corey embarked on a series of studies that laid the foundation for our present understanding of protein structure. The Peptide Bond Is Rigid and PlanarĬovalent bonds also place important constraints on the conformation of a polypeptide. The dimensions of the peptide chain are known exactly. This fixes the steric arrangement at the α-carbon atom. Stereochemistry of peptide chainsĪll proteins are made of amino acids of L-configuration. Thus, a tripeptide composed of N-terminal glutamate, a cysteine, and a C-terminal glycine is called glutamyl-cysteinyl-glycine. The naming of Peptidesįor naming peptides, the amino acid suffixes –ine (glycine), -an (tryptophan), -ate (glutamate) are changed to –yl with the exception of C-terminal amino acid. This is the chemical shorthand to write proteins. The amino acids in a peptide or protein are represented by the 3-letter or one-letter abbreviation. The C-terminal residues from its fangs and the N-terminal residues from its rattle. To represent the peptide structure, we must write in “ Rattle Snake moving representation” from left to right across the page. Incidentally, protein biosynthesis also starts from the N-terminal amino acid.  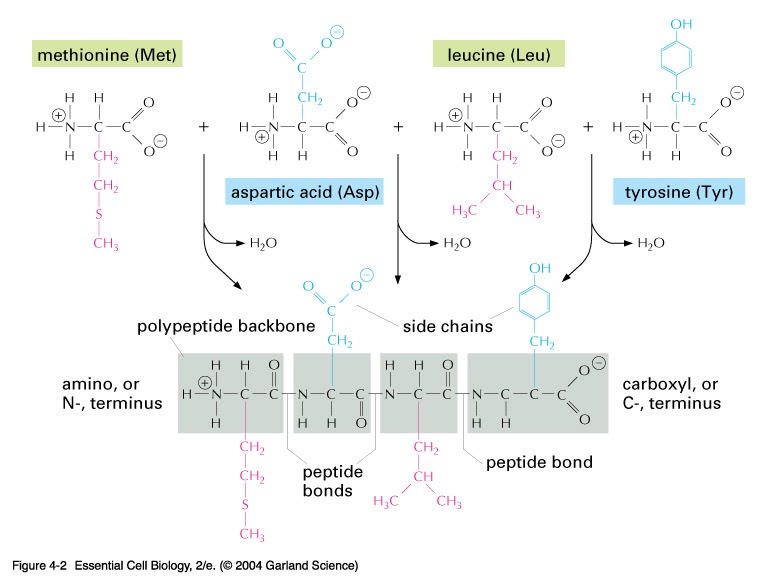
The amino acid sequence is read from the N-terminal end to the C-terminal end. Writing of Peptide structures (or) N and C-terminalsĬonventionally, the peptide chains are written with the free amino end (N-terminal residue) at the left, and the free carboxyl end (C-terminal residue) at the right. Both –C=O and –NH groups of peptide bonds are planar and are involved in hydrogen bond formation. It generally exists in trans-configuration. The peptide bond is rigid and planar, with a partial double bond in character. The peptide bond shows a partial double bond character.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
